Como a maioria dos fabricantes de medicamentos não produz produtos em embalagens que atendam às necessidades do mercado, as farmácias hospitalares tiveram que desenvolver uma verdadeira linha de produção industrial.
A maioria dos medicamentos que chega à unidade de assistência não está adequada para ser distribuída e administrada de maneira segura. Isso acontece porque cerca de 70% do que a indústria farmacêutica produz é voltado para o varejo, não para a realidade hospitalar.
Essa realidade fez com que o Cristália investisse na inovação das embalagens primárias dos seus produtos voltados ao mercado hospitalar, adotando as inovações incrementais em suas embalagens:
A ANVISA tomou providências sugerindo algumas ações, como a RDC 71, que em um de seus capítulos e anexos sugere a implementação de etiquetas coloridas, nas quais as cores estão relacionadas à classe terapêutica do produto. Embora interessante e viável, essa conduta não é obrigatória. Mesmo assim, o Cristália decidiu adotar a medida e novamente inovou!
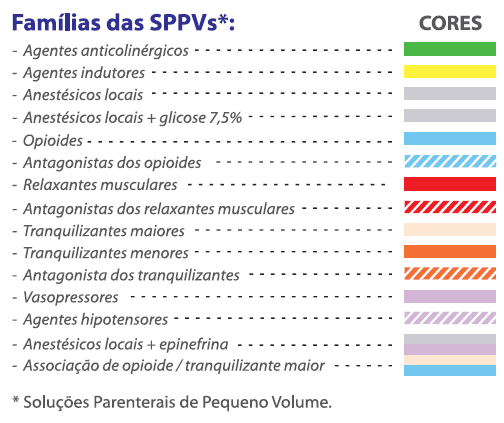
O Cristália buscou no mercado internacional um produtor com capacidade de fornecer ampolas com anéis coloridos e alinhados à sugestão da RDC 71.
Desde então, o Cristália disponibiliza ao mercado ampolas e frascos ampolas que possuem não só os rótulos coloridos conforme orientação sugerida na RDC 71, mas também ampolas com anéis nas mesmas cores, gravadas, com a “sigla / abreviatura” referente ao fármaco ali contido.